Your Position>:homepage ACADEMIC ACADEMIC ACTIVITIES
ACADEMIC ACTIVITIES
Faculty from Huzhou University Publishes Research in Internationally Renowned Journal Journal Colloid and Interface Science
[作者]: [来源]:School of Sciences [时间]:2025-12-30 9:55:29 [阅读次数]:11Recently, Associate Professor Zang Xufeng from the School of Science at Huzhou University published a research article titled “Engineering interfacial chemistry and solvation structure via a novel ionic additive for 5 V-cFlass LiNi0.5Mn1.5O4 batteries” in the internationally recognized chemistry journal Journal of Colloid and Interface Science (Chinese Academy of Sciences Q1, IF: 9.4), with Huzhou University listed as the first affiliation.

Lithium metal batteries (LMBs), as advanced energy storage systems, have been widely applied in portable electronic devices, electric vehicles, and grid energy storage. To achieve higher energy density, research has focused on developing cathode materials with higher operating voltage and capacity, such as nickel-rich layered oxides. Cobalt-free spinel lithium nickel manganese oxide (LNMO) stands out as a strong candidate for next-generation cathodes due to its high-voltage platform (>4.6 V vs. Li/Li+), near-theoretical capacity of 147 mAh/g, and high energy density of 650 Wh/kg when paired with a lithium metal anode with ultra-high theoretical specific capacity (3860 mAh/g). However, its practical application is severely hindered by rapid capacity decay caused by electrolyte decomposition, transition metal dissolution, and lithium dendrite growth. Therefore, there is an urgent need for a suitable novel bifunctional electrolyte additive.
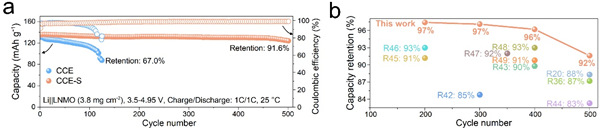
The study proposes a new bifunctional electrolyte additive —— 1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2] octane bis(tetrafluoroborate) (Selectfluor), which contains a tetrafluoroborate anion (BF4–) and a unique ammonium cation (abbreviated as [Cl-F-DA]2+). The BF4– anion preferentially oxidizes on the LNMO cathode and reduces on the lithium anode, forming an interphase rich in electron-blocking LiF and ion-conductive LiBO2. Unlike conventional additives, Selectfluor can also effectively regulate solvation structure. The BF4– anion coordinates directly with Li+ and promotes the formation of anion-derived interphases, while the [Cl-F-DA]2+ cation competes for Li+ and electrostatically attracts free solvent molecules, thereby reducing solvent coordination. Li||LNMO full cells with 1% Selectfluor in the electrolyte retained 91.6% of their capacity after 500 cycles, far exceeding the control sample (67.0% after 125 cycles). This strategy provides key insights for designing high-voltage electrolytes and accelerating the commercialization of LNMO.
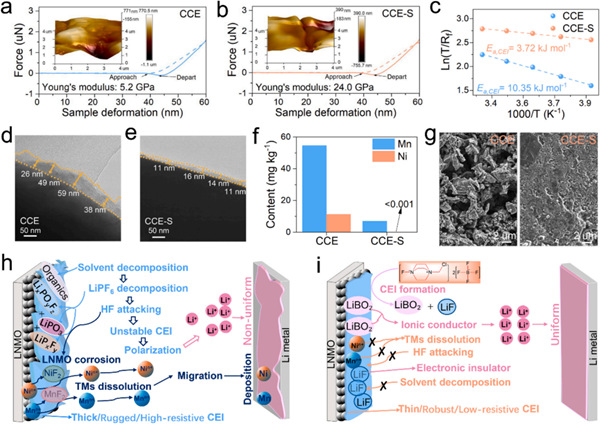
The outstanding performance improvement of Selectfluor can be attributed to two key aspects. First, it modulates Li+ solvation structure to enhance anion participation and builds a robust LiF–LiBO2 hybrid interphase on both electrodes. Second, the resulting LiF–LiBO2 electrode/electrolyte interface exhibits complementary synergistic effects: LiF provides excellent electronic insulation and mechanical strength, while LiBO2 ensures good ionic conductivity and low Li+ migration barriers. This composite structure effectively suppresses side reactions and guides uniform lithium deposition. This work offers a novel approach for developing high-performance, low-cost electrolytes for high-voltage lithium metal batteries, with broad applicability.
Article link: https://www.sciencedirect.com/science/article/pii/S0021979725031340?dgcid=author
Address: No. 759, East 2nd Road, Huzhou, China(313000)
telephone: 0572-2322746 technical support: Pioneer


