Your Position>:homepage ACADEMIC ACADEMIC ACTIVITIES
ACADEMIC ACTIVITIES
Faculty Members from Huzhou University Publish Breakthrough Research in Advanced Functional Materials
[作者]: [来源]:Office of Research & Technology, School of Engineering [时间]:2025-06-16 17:38:49 [阅读次数]:11Recently, Professor Guangliang Chen and his team from the Dept of Materials Science and Engineering, School of Engineering, Huzhou University, published a research article titled “Activating the Microsized Cubic Co?O? with Doping Y?O? Phase Inlaid on the Plasma-Treated Soft Carbon Fiber for a Robust Electrocatalytic Alkalized-Seawater Splitting” in Advanced Functional Materials (IF: 18.5), a leading journal in materials science (Q1, CAS Category). The study marks a significant breakthrough in developing efficient, stable, and cost-effective electrocatalysts for seawater electrolysis-based hydrogen production.
Hydrogen (H?) is widely recognized as one of the most promising clean energy alternatives to fossil fuels, particularly when produced via water electrolysis powered by renewable energy. Due to its advantages—such as high purity and zero emissions—electrochemical water splitting is considered one of the most reliable methods of hydrogen generation. Overall water splitting (OWS) involves two half-reactions: the hydrogen evolution reaction (HER) at the cathode and the oxygen evolution reaction (OER) at the anode. However, the slow kinetics associated with multi-electron transfer processes and the high cost of noble-metal catalysts (e.g., Pt for HER and RuO?/IrO? for OER) limit large-scale applications. Additionally, the presence of various cations and anions in seawater tends to poison and deactivate conventional catalysts, posing another major challenge for seawater electrolysis.

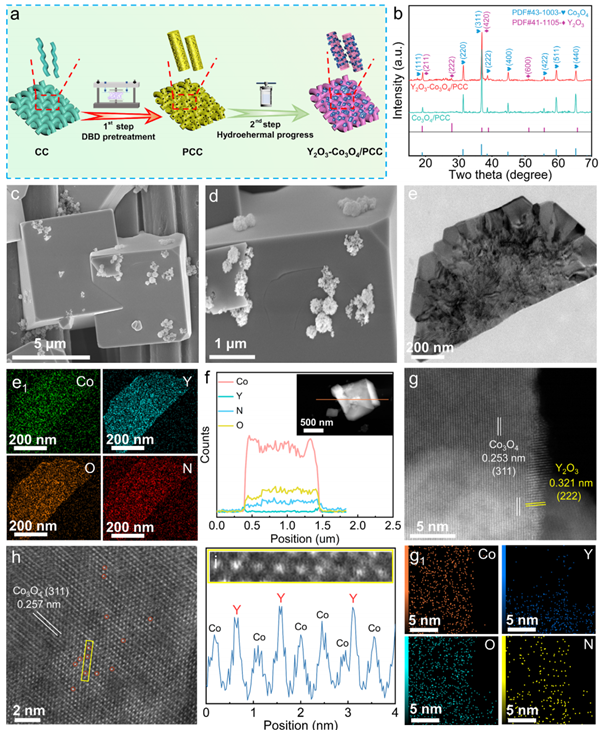
To address these issues, the study introduces a novel bifunctional non-noble metal catalyst—Y?O?-doped cubic Co?O? synthesized on plasma-treated carbon cloth (PCC). The resulting Y?O?-Co?O?/PCC catalyst exhibits outstanding bifunctional electrocatalytic performance for seawater splitting. At the optimal synthesis temperature of 250?°C, the catalyst forms abundant heterogeneous interfaces, which optimize active sites and enhance electron transfer. Under a high current density of 1000?mA?cm?? (j????), the catalyst achieves low overpotentials of 301?mV (in alkaline solution) and 346?mV (in seawater) for HER. When integrated into an anion exchange membrane water electrolyzer (AEMWE), the catalyst enables low cell voltages of 2.11?V (alkaline) and 2.47?V (alkaline seawater) at j????.
This work highlights the synergistic effects of plasma treatment and heterointerface engineering in enhancing both activity and stability of Co?O?-based catalysts. It provides theoretical and experimental support for engineering high-performance, non-noble-metal-based oxide soft electrodes for seawater electrolysis. The findings are expected to contribute to the industrial application of renewable hydrogen production and support China’s national energy strategy and dual-carbon (“carbon peak and neutrality”) goals.
Link to the article: https://doi.org/10.1002/adfm.202508158
Address: No. 759, East 2nd Road, Huzhou, China(313000)
telephone: 0572-2322746 technical support: Pioneer


